
Retatrutide Dosage & Titration Protocol: Week-by-Week Schedule
Share
Retatrutide Dosage & Titration Protocol: The Week-by-Week Schedule
- TRIUMPH Phase 3 protocol: Start at 0.5 mg/week, escalate every 4 weeks to reach 12 mg by week 21
- Full titration schedule: 0.5 → 1 → 2 → 4 → 8 → 12 mg (one dose increase every 4 weeks)
- Once weekly subcutaneous injection — same day each week
- Reconstitute 10 mg vial in 2 mL bacteriostatic water = 5 mg/mL concentration
- GI side effects peak at the 4 mg step — slow down or pause here if needed
- Not FDA-approved; research compound only in UAE
Getting the retatrutide dosage wrong is the single most common reason researchers experience intolerable side effects and abandon the protocol early. The GI burden of retatrutide — the nausea, vomiting, and discomfort — is almost entirely a function of how quickly the dose is escalated, not of the compound itself at maintenance.
This guide presents the complete TRIUMPH Phase 3 titration schedule, injection technique, reconstitution method, and a practical framework for managing side effects at each dose step.
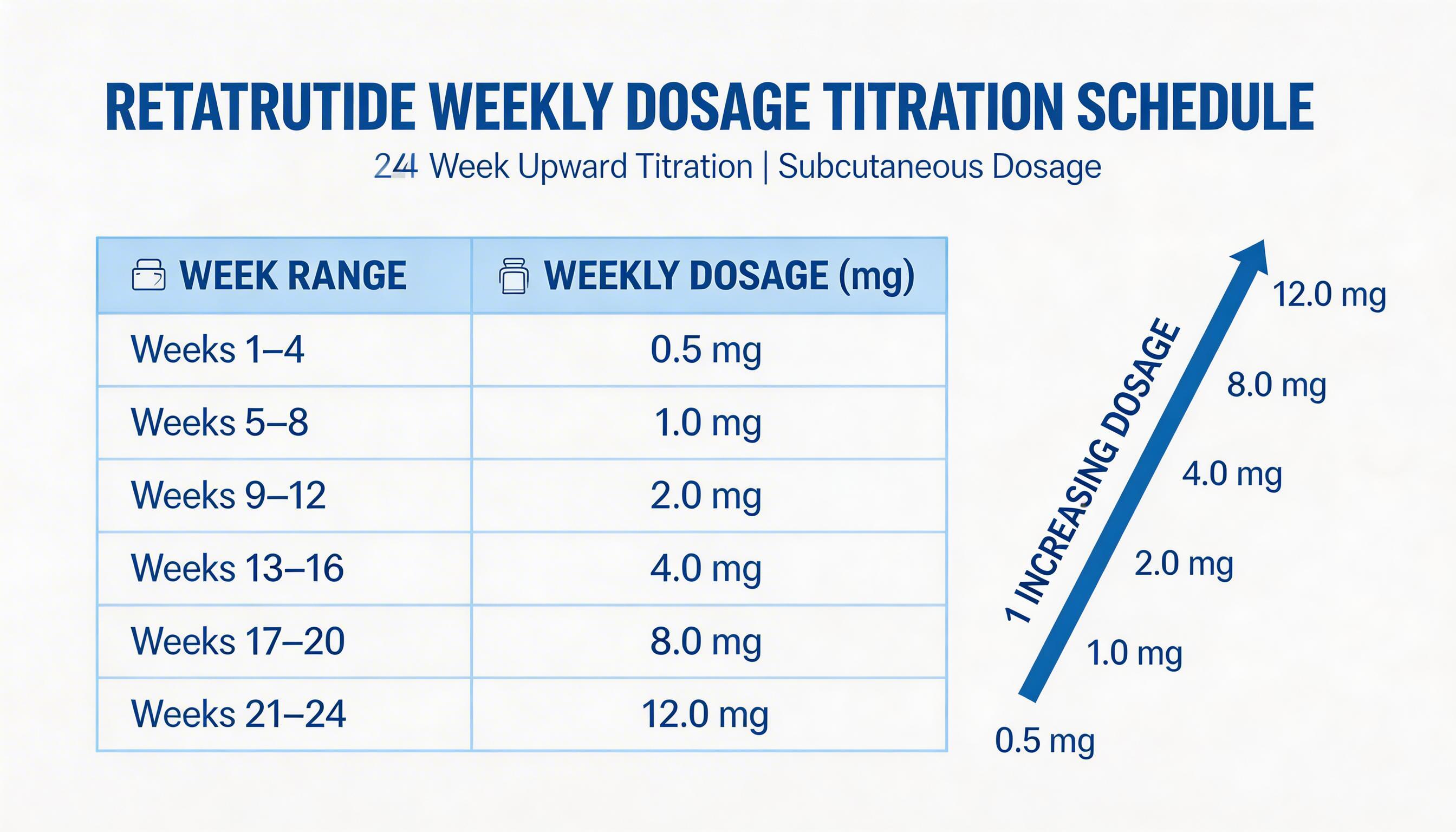
The Official TRIUMPH Titration Schedule
The TRIUMPH Phase 3 trial — Eli Lilly's pivotal programme for retatrutide (LY3437943) — established the most validated dosing protocol currently available for this triple agonist. The protocol uses a conservative slow-titration approach with dose increases every 4 weeks, which dramatically reduces the rate of GI adverse events compared to faster escalation.
| Weeks | Weekly Dose | Phase | Notes |
|---|---|---|---|
| 1–4 | 0.5 mg | Initiation | Minimal GI burden; appetite suppression begins subtly |
| 5–8 | 1.0 mg | Escalation | Some appetite reduction; mild nausea possible at dose change |
| 9–12 | 2.0 mg | Escalation | Noticeable weight loss begins; GI symptoms if eating patterns unchanged |
| 13–16 | 4.0 mg | Escalation | GI peak risk — most common step where tolerance breaks down; pause here if needed |
| 17–20 | 8.0 mg | Escalation | Strong metabolic effects; significant weight loss acceleration; fatigue possible |
| 21+ | 12.0 mg | Maintenance | Maximum efficacy dose; GI symptoms typically stabilised by this point |
| Optional | 15.0 mg | Extended | TRIUMPH allowed further escalation to 15 mg in tolerant participants |
Flexible Maintenance Doses
Not every researcher needs to reach 12 mg. The TRIUMPH protocol allowed dose flexibility — participants could remain at lower maintenance doses if higher doses were not tolerated. Clinically meaningful weight loss occurs at every step:
- 4 mg maintenance: ~14–16% body weight reduction at 48 weeks
- 8 mg maintenance: ~20–22% body weight reduction at 48 weeks
- 12 mg maintenance: ~24% body weight reduction at 48 weeks (TRIUMPH-4: 28.7% at 68 weeks)
Injection Technique
Retatrutide is administered as a once-weekly subcutaneous injection. The key rules:
- Frequency: Once every 7 days — same day each week for consistency
- Injection sites: Abdomen (preferred), upper thigh, or upper outer arm
- Site rotation: Rotate within the same region each week to prevent lipodystrophy and injection site reactions
- Needle: 29–31G, 4–8 mm insulin syringe or pen needle
- Technique: Pinch the skin, insert at 45–90°, inject slowly, hold 5 seconds before withdrawing
- Temperature: Allow vial to reach room temperature before injecting — cold solution increases injection site discomfort
Reconstituting Retatrutide from a Research Vial
Research-grade retatrutide arrives as a lyophilised (freeze-dried) white powder. Before use, it must be reconstituted with bacteriostatic water.
Standard Reconstitution (10 mg vial)
- Allow the vial to reach room temperature (15–20 minutes)
- Wipe the top of the vial and bacteriostatic water vial with an alcohol swab
- Draw 2 mL of bacteriostatic water into a syringe
- Inject the water slowly down the side of the vial — not directly onto the powder
- Do NOT shake — gently swirl or roll the vial until the powder dissolves completely
- The solution should be clear and colourless. Slight cloudiness can normalise after a few minutes
- Store reconstituted vial at 2–8°C (refrigerator). Do not freeze
Dosing Reference (10 mg vial in 2 mL = 5 mg/mL = 5,000 mcg/mL)
| Target Dose | Volume to Draw | Insulin Syringe Units |
|---|---|---|
| 0.5 mg | 0.1 mL | 10 units |
| 1.0 mg | 0.2 mL | 20 units |
| 2.0 mg | 0.4 mL | 40 units |
| 4.0 mg | 0.8 mL | 80 units |
| 8.0 mg | 1.6 mL | 160 units (use 3 mL syringe) |
| 12.0 mg | 2.4 mL* | Requires second vial at this concentration |
*For the 12 mg dose, consider reconstituting a 10 mg vial in 1 mL to achieve 10 mg/mL concentration, making 12 mg = 1.2 mL.
Side Effect Management at Each Dose Step
The most important concept in retatrutide dosing is that GI side effects are almost always dose-escalation phenomena, not stable-state phenomena. Once the body adapts to a dose level over 4 weeks, the nausea and discomfort typically reduce substantially before the next increase.
Weeks 1–8 (0.5–1 mg) — Low Risk
Most researchers tolerate the initiation phase well. Appetite suppression is subtle. If any nausea occurs, it is typically mild and brief. Focus: eat smaller meals, avoid high-fat foods on injection day.
Weeks 9–12 (2 mg) — First Real Effects
Weight loss becomes noticeable. Some researchers experience meaningful nausea at this step. Management: eat 4–5 small meals instead of 3 large ones; avoid eating within 2 hours of injection.
Weeks 13–16 (4 mg) — Critical Tolerance Test
This is the step with the highest dropout risk in research protocols. The jump from 2 mg to 4 mg represents a 100% dose increase and triggers the strongest GI response of the entire protocol.
- If tolerable: Continue to week 17
- If severe: Remain at 4 mg for 8 weeks total instead of 4 before proceeding
- Management: Ondansetron (anti-nausea), ginger tea, small frequent meals, maximum hydration
Weeks 17–20 (8 mg) — Acceleration Phase
Strong weight loss acceleration. Some researchers experience mild fatigue as energy partitioning shifts significantly. Heart rate increases of 5–10 bpm are normal at this stage. Ensure adequate electrolyte intake.
Weeks 21+ (12 mg) — Maintenance
GI symptoms, which peaked during escalation, typically stabilise substantially by the time 12 mg is reached. The body has had 20 weeks to adapt. The key side effect unique to retatrutide at this stage is dysaesthesia — an unpleasant skin sensation affecting approximately 20% of researchers at the 12 mg dose. It is usually mild and rarely requires dose reduction.
The golden rule of retatrutide dosing
It is always better to escalate more slowly than the standard protocol suggests rather than push through severe side effects. Staying at 4 mg for 8 weeks instead of 4 weeks before advancing to 8 mg does not meaningfully affect long-term outcomes — but abandoning the protocol at week 14 due to intolerable nausea does.
Frequently Asked Questions
What is the starting dose of retatrutide?
The TRIUMPH Phase 3 protocol starts at 0.5 mg once weekly for the first 4 weeks. Some earlier Phase 2 trials started at 1–2 mg, but the 0.5 mg start is now the most validated approach for tolerability.
How long does it take to reach 12 mg retatrutide?
Following the TRIUMPH schedule: 20–24 weeks (5–6 months), with dose increases every 4 weeks across 5 escalation steps.
Can you stay at a lower dose?
Yes. Significant weight loss occurs at 4 mg (~14–16%) and 8 mg (~20–22%). Not all researchers need or tolerate 12 mg. The protocol supports flexible maintenance at any dose level.
How do you reconstitute retatrutide?
Add 2 mL bacteriostatic water to a 10 mg vial = 5 mg/mL. Inject water slowly down the vial wall, gently swirl, do not shake. Store at 2–8°C.
What should you do if side effects are too severe?
Pause escalation and hold the current dose for an additional 4 weeks, or step back one level. Most GI effects resolve with stable dosing over 8–12 weeks. Anti-nausea medication, small meals, and hydration all help.
Where can I buy retatrutide in the UAE?
Research-grade retatrutide is available in the UAE through specialist peptide suppliers at AED 700–1,500 per 10 mg vial. See our Buy Retatrutide UAE guide for current pricing and sourcing information.
Research-Grade Retatrutide in UAE
CoreSup supplies verified research-grade retatrutide with Certificate of Analysis for UAE researchers.
View Retatrutide











