
PT-141 First-Time User Guide — What Beginners Need to Know
Share
PT-141 First-Time User Guide: What Beginners Need to Know
Reviewed by the CoreSup Research Team · Based on FDA prescribing information, Phase 3 RECONNECT trial protocols, and peer-reviewed peptide administration guidelines · Updated March 2026
PT-141 (bremelanotide) is a well-studied research compound with an FDA approval trail and published Phase 3 data — but that doesn't mean the first experience is without uncertainty. Nausea affects roughly 40% of users at the standard dose, onset timing is specific, and reconstitution from lyophilised powder requires correct technique. Getting these details right before the first use is the difference between a controlled, informative research experience and an unnecessarily uncomfortable one.
This guide covers everything a first-time PT-141 researcher in the UAE needs to know: what to prepare, how to reconstitute and measure doses correctly, the injection technique, what to expect in the first hour, how to manage side effects, and the frequency limits that matter for sustained effectiveness.
TL;DR: Start at 0.5–1.0 mg to test tolerance (not the full 1.75 mg FDA dose). Reconstitute with bacteriostatic water. Inject subcutaneously into the abdomen 45–60 minutes before anticipated activity. Nausea peaks at ~60 minutes and resolves within 2 hours for most users. Effects on desire last 6–12 hours. Maximum: 1 dose per 24 hours, 8 per month.
First time with PT-141? Read the background in our Complete PT-141 Guide. For side effect management specifics, see our PT-141 Side Effects Guide.
Pre-Use Checklist: What to Do Before Your First Dose
Preparation eliminates most first-time complications. Before administering PT-141 for the first time, confirm the following:
| Item | Details | Status |
|---|---|---|
| Medical review | No cardiovascular conditions; no uncontrolled hypertension; not taking naltrexone | Confirm with physician |
| Supply check | PT-141 vial (lyophilised); bacteriostatic water; insulin syringes (27–31G); alcohol swabs | All items present |
| Storage verified | Unreconstituted powder stored at 2–8°C (refrigerator) away from light | Check vial condition |
| Certificate of analysis | Lab test confirming identity and purity of your specific batch | Reviewed |
| Dose decided | First-time dose: 0.5–1.0 mg (not the full 1.75 mg) to assess tolerance | Planned |
| Timing planned | Administration 45–60 min before intended activity; first use ideally in the evening | Scheduled |
| Light meal | Eat a light meal or snack 1–2 hours before; high-fat meals delay onset and worsen nausea | Planned |
| Nausea plan | OTC anti-nausea medication (e.g., ondansetron if available, or ginger tablets) on hand | Available |
How to Reconstitute PT-141 Powder
Most PT-141 available for research comes as a lyophilised (freeze-dried) white powder in a sealed glass vial. This must be reconstituted with bacteriostatic water (BW) before injection. Do not use regular sterile water (it doesn't contain the preservative that allows multi-dose storage) and do not use tap water.
Step 1 — Calculate your concentration
Decide what concentration you want. A common starting point for a 10 mg vial: add 2 mL of bacteriostatic water to create a 5 mg/mL solution. This means each 0.1 mL drawn equals 0.5 mg of PT-141. For a 1.75 mg dose: draw 0.35 mL. For a first-time 1.0 mg dose: draw 0.2 mL.
Step 2 — Clean both vial tops with an alcohol swab
Wipe the rubber septum of both the PT-141 vial and the bacteriostatic water vial with fresh alcohol swabs. Allow to air-dry for 10 seconds.
Step 3 — Draw the bacteriostatic water
Using a fresh insulin syringe, draw the calculated volume of BW (e.g., 2 mL requires two 1 mL syringe fills if using a 1 mL syringe). Insert the needle at an angle through the BW vial septum and draw slowly.
Step 4 — Add BW to the PT-141 vial
Insert the needle through the PT-141 vial septum and direct the BW slowly down the inside glass wall of the vial — not directly onto the powder cake. This prevents foaming. Inject slowly and steadily.
Step 5 — Gently swirl to dissolve
Gently roll the vial between your palms in a circular motion. Do not shake — shaking can cause peptide degradation. The solution should become clear within 30–60 seconds. If undissolved particles remain after 2 minutes, continue gentle swirling. Store reconstituted solution at 2–8°C (refrigerator). Use within 28 days of reconstitution.
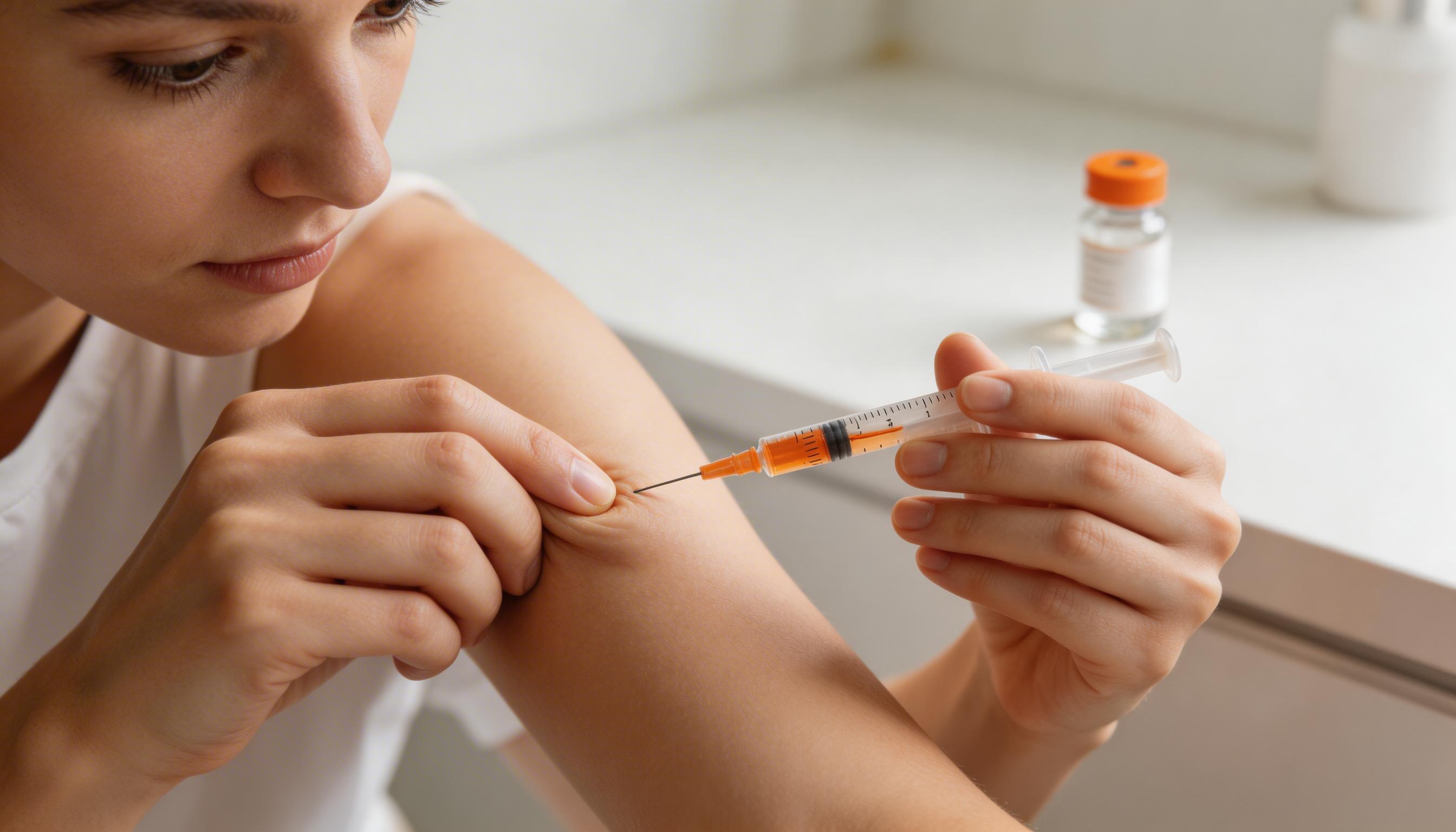
Injection Technique
Site Selection
PT-141 is a subcutaneous injection — into the layer of fatty tissue just beneath the skin, not into muscle (intramuscular) and not into the vein (intravenous). Best sites: the abdomen (pinch a fold of skin at least 2 inches from your navel), the outer thigh, or the upper arm. The abdomen is the most common choice due to predictable subcutaneous fat depth. Rotate injection sites with each use to avoid lipodystrophy (tissue changes at a repeatedly injected site).
Needle Selection
Use a 27–31 gauge, 6–8 mm (¼") insulin syringe. These are short, thin needles that minimise discomfort and are correctly sized for subcutaneous injection. Longer needles risk intramuscular injection, which would alter absorption kinetics.
Technique
- Wash hands thoroughly. Swab the chosen injection site with an alcohol swab and allow to dry.
- Draw the calculated dose volume from the reconstituted PT-141 vial using a fresh syringe.
- Pinch a fold of skin (about 1 inch across) between thumb and forefinger.
- Insert the needle at a 45–90 degree angle in one smooth motion.
- Release the skin pinch, then slowly depress the plunger over 3–5 seconds.
- Withdraw the needle and apply gentle pressure with a clean swab. Do not rub.
- Dispose of the needle safely in a sharps container.
What to Expect in the First Hour
The first hour after a PT-141 injection is when all the primary pharmacological events occur — and knowing what's normal prevents unnecessary concern.
Minutes 15–30: Absorption Phase
The compound begins absorbing from the subcutaneous depot. Some users notice very early warmth at the injection site or mild systemic warmth. Most people notice nothing in this window.
Minutes 30–60: Onset of Primary Effects
This is when most users first feel PT-141's characteristic effects. Flushing (warmth and redness in the face, neck, and chest) is common — approximately 20% of users in the RECONNECT trials reported it. This is a normal vascular response and is not a cause for concern. If nausea is going to occur, it typically begins here. The increase in sexual desire — the primary intended effect — also begins in this window. Many users report a mental "shift" toward sexual thoughts, heightened physical awareness, and increased sensitivity. In men, partial or full erections may occur spontaneously.
The 60-Minute Mark (Peak)
This coincides with Tmax (peak plasma concentration). Peak effects. If nausea is present, this is typically when it is most intense. Most nausea resolves within the next 30–60 minutes without intervention. Lying down during nausea helps. If anti-nausea medication was planned (e.g., ondansetron or domperidone), taking it around the 45-minute mark pre-emptively — before nausea peaks — is more effective than waiting for symptoms to appear.
Dosing Guidance for First-Time Use
| Session | Dose | Purpose |
|---|---|---|
| First use | 0.5 mg | Tolerance test — assess nausea sensitivity and individual response |
| Second use (3–7 days later) | 1.0 mg | Mid-range dose — typically produces clear effects with lower nausea risk than full dose |
| Third use onwards | 1.75 mg (or maintain at 1.0 mg) | Full FDA-studied dose — appropriate once individual response and tolerance established |
Starting low is not a sign of under-dosing — it's a sign of methodical research. The difference in effect quality between 1.0 mg and 1.75 mg is real but incremental. For many users, 1.0–1.25 mg delivers satisfactory results with substantially lower nausea rates than the full 1.75 mg dose.
Ready to Start Your PT-141 Research?
CoreSup supplies lab-verified PT-141 (bremelanotide) with full certificates of analysis — delivered across the UAE with discretion and speed.
View PT-141 at CoreSupFrequently Asked Questions
What dose should I start with for PT-141?
First-time researchers frequently start at 0.5–1.0 mg to assess individual tolerance, particularly for nausea sensitivity. The FDA-approved dose is 1.75 mg, but starting lower reduces nausea risk. Once tolerance is established, dose can be titrated upward.
What happens in the first hour after taking PT-141?
Within 45–60 minutes: mild warmth, flushing, and increased sense of arousal. Nausea, if it occurs, typically begins 30–60 minutes post-injection and peaks around 60 minutes. Most nausea resolves within 1–2 hours. The primary effect — increased sexual desire — also begins in this window and can last 6–12 hours.
How do you reconstitute PT-141 powder?
Add bacteriostatic water (not sterile water) to the lyophilised powder vial. For a 10 mg vial: add 2 mL BW to create a 5 mg/mL solution. Inject the BW slowly down the vial's inner wall, gently swirl until clear, and store refrigerated for up to 28 days.
Where do you inject PT-141?
Subcutaneous (SC) injection into the fatty tissue below the skin. Best sites: abdomen (2+ inches from navel), outer thigh, or upper arm. Use a 27–31 gauge insulin syringe. Rotate sites with each use.
How often can you use PT-141?
The FDA label specifies a maximum of one dose per 24 hours and no more than 8 doses per month. Research protocols typically follow similar spacing to avoid receptor desensitisation.












