
PT-141 vs Kisspeptin — Which Peptide for Sexual Function in the UAE?
Share

PT-141 vs Kisspeptin: Which Peptide for Sexual Function in the UAE?
Reviewed by the CoreSup Research Team · Based on FDA clinical data, Imperial College London kisspeptin research, and peer-reviewed neuroendocrinology literature · Updated March 2026
PT-141 and kisspeptin both influence sexual function, but through mechanisms so different that comparing them is like comparing a light switch to a generator. PT-141 flips the arousal switch directly in the brain's desire circuits. Kisspeptin operates earlier in the chain — it's the signal that tells the hypothalamus to start the hormonal cascade that eventually produces testosterone and estrogen, which then sensitise those same brain circuits. Understanding which approach is more relevant to a specific research question determines which compound is the more appropriate choice.
TL;DR: PT-141 (MC3R/MC4R agonist) produces acute sexual desire directly. Kisspeptin (GPR54 agonist) stimulates GnRH → LH/FSH → testosterone/estrogen upstream. PT-141 has Phase 3 FDA approval; kisspeptin is in Phase 1/2 research. If desire is the acute target with an intact hormonal system → PT-141. If the hormonal axis needs upstream support → kisspeptin. They are complementary, not competing.
For PT-141 background, see the Complete PT-141 Guide. For hormone and testosterone interactions, see PT-141 and Testosterone.
What Is Kisspeptin?
Kisspeptin is a naturally occurring neuropeptide produced by kisspeptin neurons (also called KNDy neurons — for kisspeptin/neurokinin B/dynorphin) in the arcuate nucleus and anteroventral periventricular nucleus (AVPV) of the hypothalamus. It was discovered in 1996 at Hershey Medical Center and initially named "metastin" for its role in suppressing cancer metastasis — its role in reproduction was only identified in 2003, when researchers found that loss-of-function mutations in the kisspeptin receptor (GPR54, now also called KISS1R) caused hypogonadotropic hypogonadism (a condition where the body fails to produce adequate sex hormones).
Kisspeptin's primary reproductive function is as the "gatekeeper" of the HPG axis. It binds to GPR54 receptors on GnRH neurons in the hypothalamus, triggering pulsatile GnRH release. GnRH travels to the anterior pituitary where it stimulates LH and FSH secretion. LH then acts on the gonads — the testes (in men, to produce testosterone) and ovaries (in women, to produce estrogen and progesterone). Kisspeptin is therefore the upstream regulator of the entire sex hormone system.
Mechanism Comparison: Two Different Entry Points
The core pharmacological difference between PT-141 and kisspeptin is the point at which they intervene in the sexual desire pathway:
| Feature | PT-141 (Bremelanotide) | Kisspeptin |
|---|---|---|
| Target receptor | MC3R, MC4R (melanocortin) | GPR54 / KISS1R (kisspeptin) |
| Where it acts | Hypothalamic arousal centres (PVN, MPOA) — direct central effect | Hypothalamic GnRH neurons — upstream hormonal axis |
| Primary mechanism | Direct activation of sexual desire/arousal circuits | Triggers GnRH → LH/FSH → testosterone/estrogen cascade |
| Effect type | Acute (onset 45–60 min, duration 6–12 hrs) | Acute LH pulse (within minutes); sustained hormonal effects with ongoing use |
| FDA status | Approved (Vyleesi, 2019) for HSDD in women | No FDA approval; Phase 1/2 research |
| Clinical evidence | Phase 3 (RECONNECT, 1,247 participants) | Phase 1/2 (small trials, primarily Imperial College London) |
| Half-life | ~2.7 hours (FDA label) | Kisspeptin-10: ~1.7 min; kisspeptin-54: ~28 min (IV infusion) |
| Administration | Subcutaneous injection, as-needed | IV infusion (research), SC injection (experimental) |
| Affects testosterone | No direct effect | Yes — stimulates LH → testosterone increase |
| Affects desire directly | Yes — primary mechanism | Yes — but indirectly (via hormones) and possibly directly through brain GPR54 |
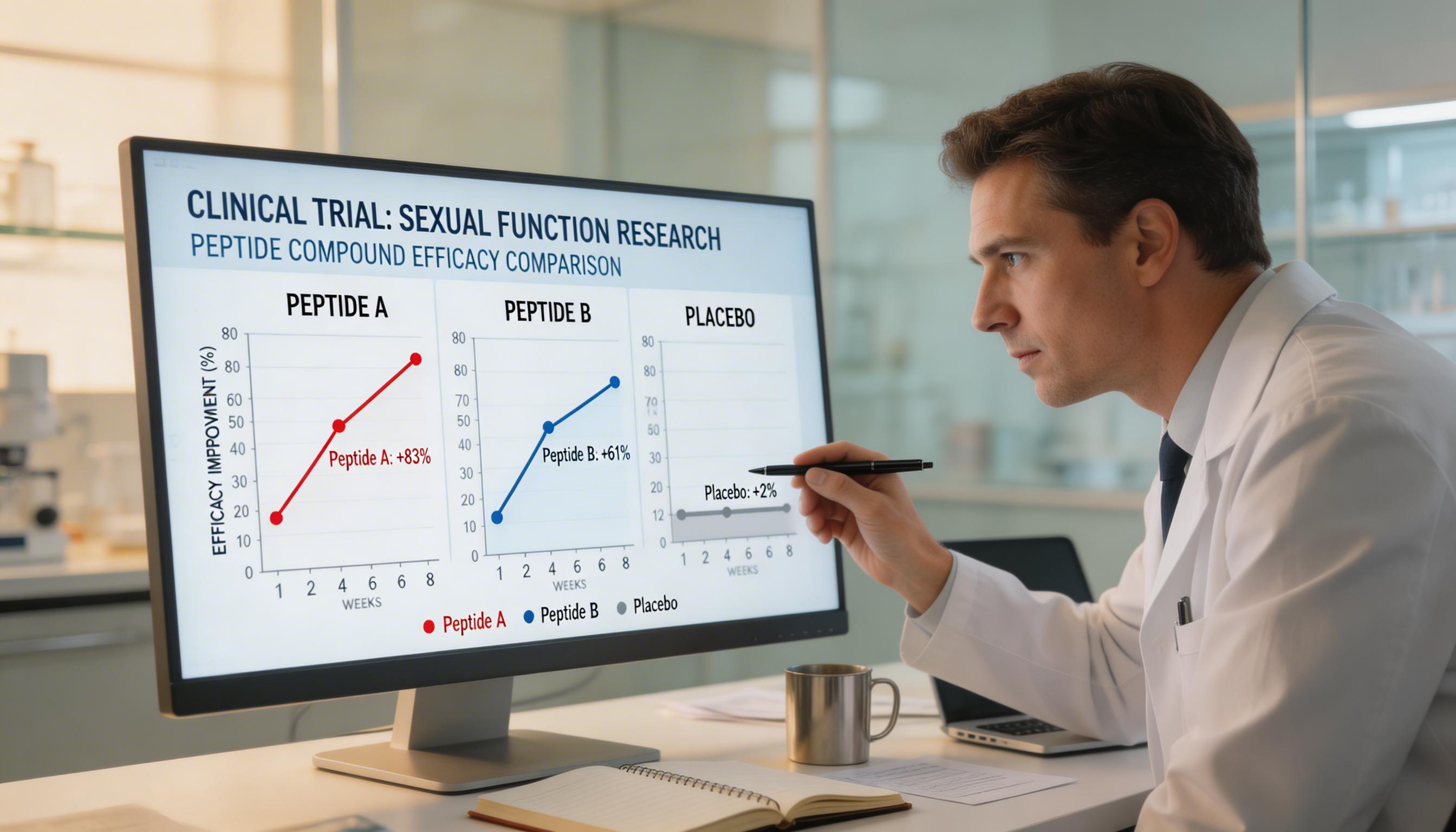
Clinical Evidence: A Clear Asymmetry
PT-141's Evidence Base
PT-141 is the only melanocortin-based sexual function compound with Phase 3 FDA-regulated trial data. The RECONNECT programme — two identical randomised, double-blind, placebo-controlled Phase 3 trials — enrolled 1,247 premenopausal women with HSDD. Both co-primary endpoints (desire improvement and distress reduction) were met with P<0.001 significance. FDA approval followed in June 2019. For men, a 2007 AUA-presented Phase 2 trial showed positive erectile responses in 33.5% of prior sildenafil non-responders. The evidence for PT-141 is among the strongest of any sexual health research compound.
Kisspeptin's Evidence Base
Kisspeptin research is at an earlier clinical stage. The landmark work from Imperial College London has produced multiple Phase 1/2 studies demonstrating that kisspeptin-54 infusion increases LH pulsatility, affects sexual brain processing, and may reduce sexual aversion in women with psychosexual dysfunction. A 2020 study enrolled women with HSDD and found kisspeptin-54 infusion improved sexual desirability scores and brain activation in reward-related regions.
However, kisspeptin has not completed Phase 3 trials for any sexual function indication. The studies are informative and scientifically compelling, but they involve small samples (typically 20–50 participants), use intravenous infusion protocols (not practical for routine use), and have not been submitted for regulatory review. The clinical gap between kisspeptin and PT-141's evidence quality is substantial.
Who Is a Candidate for Each?
PT-141 Is Appropriate When:
- The research focus is acute desire and arousal activation
- The hormonal axis is intact (testosterone and estrogen in normal range)
- The user has not responded adequately to PDE5 inhibitors (men)
- The need is for an as-needed compound with fast onset
- Evidence quality and regulatory precedent are priorities
Kisspeptin Research May Be More Relevant When:
- The research focus is on the HPG axis and testosterone regulation
- There is hypothalamic amenorrhea or hypogonadotropic hypogonadism
- The interest is in fertility-adjacent research (LH/FSH regulation)
- Long-term hormonal support rather than acute activation is the goal
- The researcher has access to an appropriate research setting (kisspeptin's current administration protocols are not as practical as PT-141)
Research-Grade PT-141 in the UAE
CoreSup supplies laboratory-verified PT-141 with certificates of analysis — purity, identity, and potency confirmed. Delivered across Dubai, Abu Dhabi, and the UAE.
View PT-141 at CoreSupFrequently Asked Questions
What is kisspeptin and how does it affect sexual function?
Kisspeptin is a neuropeptide produced in the hypothalamus that acts on GPR54 receptors to trigger GnRH release, which stimulates LH/FSH and ultimately testosterone and estrogen production. It also has direct central effects on sexual brain processing, as demonstrated in Imperial College London studies showing increased arousal-related brain activity with kisspeptin-54 infusion.
Is kisspeptin better than PT-141 for low libido?
Neither is universally better — they suit different root causes. If low libido stems from hormonal deficiency (low testosterone), kisspeptin's upstream hormonal support may be more directly relevant. If the hormonal system is intact but central arousal is deficient, PT-141's direct mechanism is more targeted. PT-141 also has significantly stronger clinical evidence (Phase 3 FDA approval vs. Phase 1/2 for kisspeptin).
Can you combine PT-141 and kisspeptin?
There is no published clinical data on combination use. Theoretically they address complementary pathways. Without controlled research data, the interaction profile and optimal dosing of combination use is unknown — this remains an open research area.
Does kisspeptin have fewer side effects than PT-141?
Kisspeptin's Phase 1/2 studies suggest a mild side effect profile. PT-141 has well-characterised side effects from large trial populations (nausea 40%, flushing 20.3%). Kisspeptin's longer-term safety profile is less well-defined due to smaller study populations.












